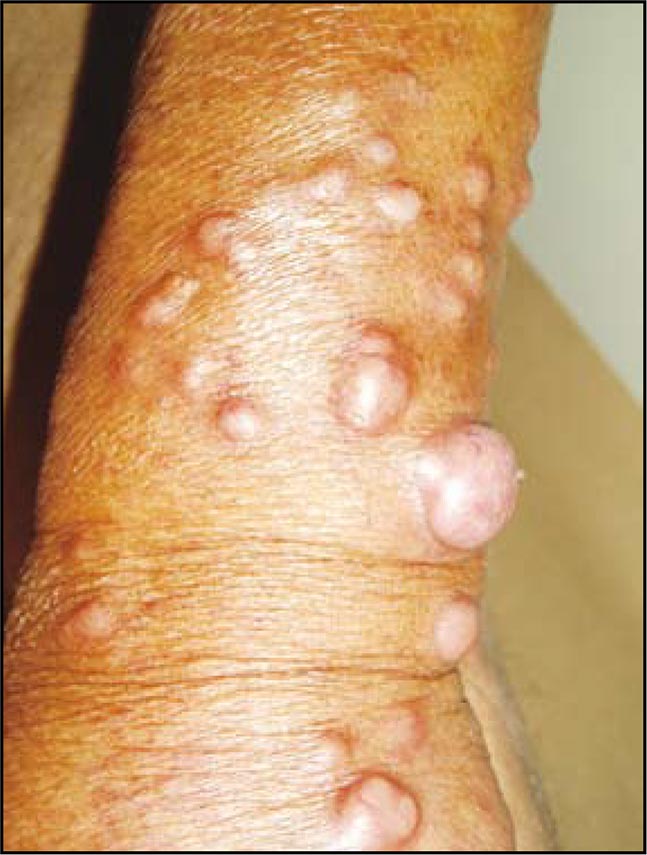
Leprosy
Keywords:
Mycobacterium leprae, piel, leprosyAbstract
Leprosy is a systemic granulomatous disease caused by Mycobacterium leprae (Hansen’s bacillus). It is transmitted from person to person and has a long incubation period (between two and six years). The disease presents polar clinical forms (the “multibacillary” lepromatous leprosy and the “paucibacillary” tuberculoid leprosy), as well as other intermediate forms with hybrid characteristics. The diagnosis, based on clinical suspicion, is confirmed through bacteriological and histopathological analyses. The Mitsuda test (intradermal reaction that is usually negative in lepromatous leprosy form and positive in the tuberculoid form) is useful to determine de the patient’s immunological state. The most commonly used drugs to treat this disease are dapsone, rifampicin and clofazimine. Early diagnosis, appropriate therapy and multidisciplinary management prevent disability and improve prognosis and quality of life in patients with Hansen disease.
Author Biographies
Ana María Rivas
RI Dermatología. Universidad Pontificia Bolivariana. Medellín, Colombia.
Luz Marina Gómez
Dermatóloga. Profesora titular, Universidad Pontificia Bolivariana. Medellín, Colombia.
References
2. Guías de promoción de la salud y prevención de enfermedades en la salud pública. Tomo III. Guía 18. Guía de atención de la lepra. 2007. p 29 URL disponible en: http://www.minproteccionsocial.gov.co/VBeContent/library/documents/DocNewsNo16159DocumentNo4194.PDF. Ingreso 4 noviembre de 2007.
3. Rodríguez G, Orozco LC. Lepra. Santafé de Bogotá: Instituto Nacional de Salud. 1996.
4. De Zubiria R, Rodríguez G. Historia de la lepra, ayer, hoy y mañana. URL disponible en: http://anm.encolombia.com/academ25161-lepra.htm ingreso 5 de noviembre de 2007.
5. Historia sanatorio Agua de Dios. URL disponible en: http://www.sanatorioaguadedios.gov.co/quienes/quienes_historia.htm Ingreso 5 de noviembre de 2007.
6. World Health Organization. Leprosy Global Situation. URL disponible en: http://www.who.int/lep/situation/en/ Ingreso 5 de noviembre de 2007.
7. Organización Panamericana de la Salud. Situación de la lepra en la Región de las Américas. URL disponible en: http://www.paho.org/Spanish/AD/DPC/CD/lep-americas.htm Ingreso 5 de noviembre de 2007.
8. Sasaki S, Takeshita F, Okuda K, Ishii N. Mycobacterium leprae and leprosy: a compendium. Microbiol Immunol. 2001;45:729-36.
9. Cho SN, Cellona RV, Villahermosa LG, Fajardo TT Jr, Balagon MV, Abalos RM, et al. Detection of phenolic glycolipid I of Mycobacterium leprae in sera from leprosy patients before and after start of multidrug therapy. Clin Diagn Lab Immunol. 2001;8:138–42.
10. Walker SL, Lockwood DN. The clinical and immunological features of leprosy. Br Med Bull. 2006;77-8:103-21.
11. De Souza FC, Marcos EV, Ura S, Opromolla PA, Nogueira ME. Comparative study between the Mitsuda test and the human leukocyte antigens in leprosy patients. Rev Soc Bras Med Trop. 2007;40:188-91.
12. Gamboa LA, Paredes M. Compromiso neurológico en la lepra. Univ med. 2003; 44 :217-223
13. Gómez JR, Moll F. Lepra: enfermedad olvidada. Situación actual y trabajo sobre el terreno. Enf Emerg. 2005;7:110-9.
14. Spiering E, De Boer T, Zulianello L, Ottenhoff TH. Novel mechanisms in the inmunopatogenesis of leprosy nerve damage: The role of Schwann cells, T cells and Mycobacterium leprae. Immunol. Cell Biol. 2000; 78: 349-55.
15. Walker SL, Lockwood DN. Leprosy. Clin Dermatol. 2007 M; 25:165-72.
16. Araújo MG. Leprosy in Brazil. Rev Soc Bras Med Trop. 2003; 36:373-8.
17. Chimenos E, Pascual M, Pinol C, Vinals H, Rodríguez de Rivera ME, López J. Lepromatous leprosy: a review and case report. Med Oral Patol Oral Cir Bucal. 2006;11: E474-9.
18. Ffytche TJ. The prevalence of disabling ocular complications of leprosy: a global study. Indian J Lepr 1998;70:49 – 9.
19. Lewallen S, Courtright P. A overview of ocular leprosy after 2 decades of multidrug therapy. Int Ophthalmol Clin. 2007;47:87-101.
20. Burstein Alva Z. La lepra (Enfermedad de Hansen). Parte II: reacciones lepróticas y secuelas. Rev Peru Med Exp Salud Publica. 2005; 22: 151-3.
21. Van Brakel WH, Khawas IB, Lucas SB. Reactions in leprosy: an epidemiological study of 386 patients in West Nepal. Lepr Rev 1994; 65:190–203.
22. Reyes Flórez O. La reacción leprosa una revisión. Derm. Venez. 2000. 38 : 85-95.
23. Fernández JL, Rangel Mayoral JF, Liso Rubio FJ. Revisión de la enfermedad de Hansen. Farm Hosp. 2004; 28: 123-9.
24. Boggild AK, Keystone JS, Kain KC. Leprosy: a primer for Canadian physicians. CMAJ. 2004 Jan 6;170:71-8.
25. Croft RP, Richardus JH, Smith WCS. Field treatment of acute nerve function impairment in leprosy using a standardized corticosteroid regimen — first year’s experience with 100 patients. Lepr Rev 1997;68:316-25.
26. Pocaterra L, Jain S, Reddy R, Muzaffarullah S, Torres O, Suneetha S, et al. Clinical course of erythema nodosum leprosum: an 11-year cohort study in Hyderabad, India. Am J Trop Med Hyg 2006;74:868–79.
27. Sarno EN, Grau GE, Vieira LM, Nery JA. Serum levels of tumour necrosis factor-alpha and interleukin-1 beta during leprosy reactional states. Clin Exp Immunol 1991;84:103- 8.
28. Sampaio EP, Duppre NC, Nery JA, Moreira AL, Sarno EN. Development of giant reaction in response to PPD skin test in lepromatous leprosy patients. Int J Lepr Other Mycobact Dis. 1993;61:205-13.
29. Manandhar R, LeMaster JW, Roche PW. Risk factors for erythema nodosum leprosum. Int J Lepr Other Mycobact Dis. 1999;67:270 -8.
30. Rijal A, Agrawal S, Agarwalla A, Lakhey M. Bullous erythema nodosum leprosum: a case report from Nepal. Lepr Rev 2004;75:177- 80.
31. Como reconocer y tratar las reacciones leprosas. Guía de aprendizaje No. 2. ILEP. URL disponible en: http://www.ilep.org.uk/documents/lg2sp.pdf Ingreso 8 de enero de 2008.
32. Helmer KA, Fleischfresser I, Kucharski-Esmanhoto LD, Neto JF, Santamaría JR. The Lucio's phenomenon (necrotizing erythema) in pregnancy. An Bras Dermatol. 2004; 79:205-10.
33. Costa IM, Kawano LB, Pereira CP, Nogueira LS. Lucio's phenomenon: a case report and review of the literature. Int J Dermatol. 2005 ;44:566-71.
34. Sehgal VN. Lucio's phenomenon/erythema necroticans. Int J Dermatol. 2005;44:602-5.
35. Rodríguez G, Pinto R, Laverde C, Sarmiento M, Riveros A, Valderrama J, et al. Recidivas postratamiento de la lepra multibacilar. Biomédica. 2004;24: 133-9.
36. WHO Expert Committee on Leprosy. 7th report. no. 874 of Technical Report series. Geneva: World Health Organization; 1998.
37. URL disponible en www.clinicalevidence.com. Fecha de ingreso 15 de Enero de 2008.
38. Girdhar BK, Girdhar A, Kumar A. Relapses in multibacillary leprosy patients: effect of length of therapy. Lepr Rev 2000;71:144–53.
39. Efficacy of single dose multidrug therapy for the treatment of single-lesion paucibacillary leprosy. Single-lesion Multicentre Trial Group. Indian J Lepr 1997;69:121-9.
40. Zodpey SP. Protective effect of bacillus Calmette Guerin (BCG) vaccine in the prevention of leprosy: a meta-analysis. Indian J Dermatol Venereol Leprol. 2007;73:86-93.
How to Cite
Downloads
Downloads
Published
How to Cite
Issue
Section
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |