Eosinophils in bullous pemphigoid: a narrative review
DOI:
https://doi.org/10.29176/2590843X.1998Keywords:
vesiculobullous skin diseases, Bullous pemphigoid, eosinophils, immunoglobulin E, pathophysiologyAbstract
Bullous Pemphigoid (BP) is an uncommon autoimmune blistering disease primarily affecting the elderly, with an incidence ranging from 2.4 to 42.8 cases per million person-years. In Colombia, it ranks among the most frequent autoimmune blistering diseases, predominantly affecting women over 70 years old. BP typically presents as tense blisters on erythematous or urticarial plaques, mainly on the trunk and flexural areas, and may involve mucous membranes in 10-30% of cases. Diagnosis is based on clinical features and paraclinical tests such as histopathology and immunofluorescence. Higher severity and mortality have been associated with elevated eosinophil counts in tissues.
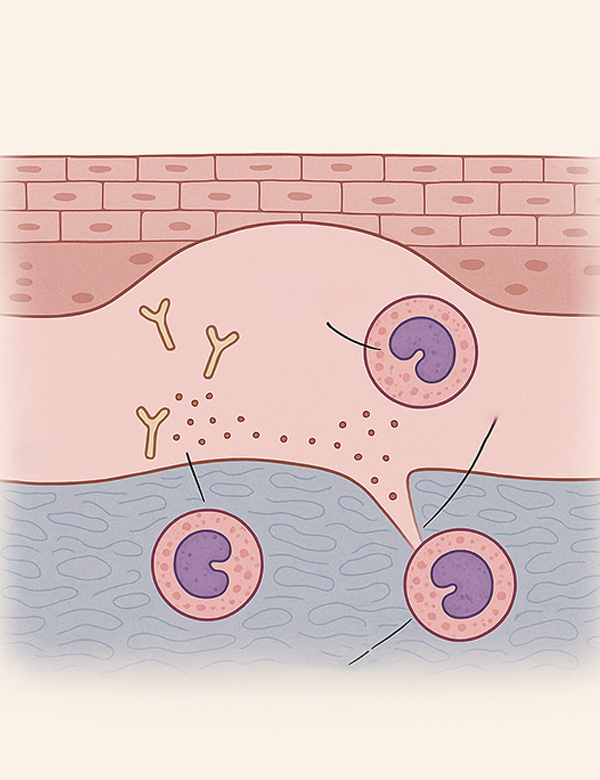
Eosinophils, normally present in low levels, can increase in allergic and infectious diseases. In BP, eosinophils play a crucial role in blister formation through degranulation, which degrades the basement membrane. BP pathogenesis involves autoantibodies against hemidesmosome components like BP180 and complement activation, which recruits eosinophils. IgE also plays a significant role in eosinophil activation.
Standard treatment includes glucocorticoids and sometimes immunosuppressive therapies. Biological options such as rituximab, omalizumab, and dupilumab have shown benefits, particularly in refractory cases. New therapies targeting eosinophils and their pathways are under study, showing promising preliminary results. Understanding the role of eosinophils in BP is leading to the development of more specific and effective treatments.
Author Biographies
Artur Moraes-L'Abbate, Pontificia Universidad Javeriana, Bogotá, Colombia
Residente de Dermatología, Pontificia Universidad Javeriana, Bogotá, Colombia
Carolina Cortés Correa, Hospital Universitario de la Samaritana, Bogotá, Colombia
Dermatóloga, Hospital Universitario de la Samaritana, Bogotá, Colombia.
References
Lu L, Chen L, Xu Y, Liu A. Global incidence and prevalence of bullous pemphigoid: A systematic review and meta-analysis. J Cosmet Dermatol 2022 -10;21(10):4818-4835.
Amber KT, Murrell DF, Schmidt E, Joly P, Borradori L. Autoimmune Subepidermal Bullous Diseases of the Skin and Mucosae: Clinical Features, Diagnosis, and Management. Clin Rev Allergy Immunol 2018 -02;54(1):26-51.
Ximena Alexandra Pinilla Garcia. Estudio de las características clínicas y epidemiológicas de los pacientes con enfermedades ampollosas autoinmunes en un hospital de tercer nivel de Bogotá Universidad Nacional de Colombia; 2015.
Zuluaga DC, Del Río DY, Úsuga YA, Aguirre-Acevedo D, Velásquez MM. Estudio retrospectivo clínico-epidemiológico de los pacientes con pénfigo y penfigoide ampolloso del Hospital Universitario San Vicente Fundación. Rev Asoc Colomb Dermatol Cir Dematol 2017;25(2):118-129.
Kayani M, Aslam AM. Bullous pemphigoid and pemphigus vulgaris. BMJ 2017 -06-08;357:j2169.
Miyamoto D, Santi CG, Aoki V, Maruta CW. Bullous pemphigoid. An Bras Dermatol 2019;94(2):133-146.
Lamberts A, Meijer JM, Jonkman MF. Nonbullous pemphigoid: A systematic review. J Am Acad Dermatol 2018 -05;78(5):989-995.e2.
Gore Karaali M, Koku Aksu AE, Cin M, Leblebici C, Kara Polat A, Gurel MS. Tissue eosinophil levels as a marker of disease severity in bullous pemphigoid. Australas J Dermatol 2021 -05;62(2):e236-e241.
Davoine F, Lacy P. Eosinophil Cytokines, Chemokines, and Growth Factors: Emerging Roles in Immunity. Front Immunol 2014 -11-10;5:570.
Marichal T, Mesnil C, Bureau F. Homeostatic Eosinophils: Characteristics and Functions. Front Med (Lausanne) 2017;4:101.
Jung Y, Rothenberg ME. Roles and regulation of gastrointestinal eosinophils in immunity and disease. J Immunol 2014 -08-01;193(3):999-1005.
Rothenberg ME, Hogan SP. The eosinophil. Annu Rev Immunol 2006;24:147-174.
Akuthota P, Wang H, Weller PF. Eosinophils as Antigen-Presenting Cells in Allergic Upper Airway Disease. Curr Opin Allergy Clin Immunol 2010 -2;10(1):14-19.
Bağcı IS, Horváth ON, Ruzicka T, Sárdy M. Bullous pemphigoid. Autoimmun Rev 2017 -05;16(5):445-455.
Liu Y, Li L, Xia Y. BP180 Is Critical in the Autoimmunity of Bullous Pemphigoid. Front Immunol 2017;8:1752.
Hertl M, Eming R, Veldman C. T cell control in autoimmune bullous skin disorders. J Clin Invest 2006 -5-1;116(5):1159-1166.
Chen R, Fairley JA, Zhao M, Giudice GJ, Zillikens D, Diaz LA, et al. Macrophages, but not T and B lymphocytes, are critical for subepidermal blister formation in experimental bullous pemphigoid: macrophage-mediated neutrophil infiltration depends on mast cell activation. J Immunol 2002 -10-01;169(7):3987-3992.
Messingham KAN, Noe MH, Chapman MA, Giudice GJ, Fairley JA. A novel ELISA reveals high frequencies of BP180-specific IgE production in bullous pemphigoid. J Immunol Methods 2009 -07-31;346(1-2):18-25.
Zone JJ, Taylor T, Hull C, Schmidt L, Meyer L. IgE basement membrane zone antibodies induce eosinophil infiltration and histological blisters in engrafted human skin on SCID mice. J Invest Dermatol 2007 -05;127(5):1167-1174.
Amber KT, Chernyavsky A, Agnoletti AF, Cozzani E, Grando SA. Mechanisms of pathogenic effects of eosinophil cationic protein and eosinophil-derived neurotoxin on human keratinocytes. Exp Dermatol 2018 -12;27(12):1322-1327.
Messingham KN, Holahan HM, Frydman AS, Fullenkamp C, Srikantha R, Fairley JA. Human Eosinophils Express the High Affinity IgE Receptor, FcεRI, in Bullous Pemphigoid. PLoS One 2014 -9-25;9(9):e107725.
de Graauw E, Sitaru C, Horn M, Borradori L, Yousefi S, Simon H-, et al. Evidence for a role of eosinophils in blister formation in bullous pemphigoid. Allergy 2017 -07;72(7):1105-1113.
Verraes S, Hornebeck W, Polette M, Borradori L, Bernard P. Respective contribution of neutrophil elastase and matrix metalloproteinase 9 in the degradation of BP180 (type XVII collagen) in human bullous pemphigoid. J Invest Dermatol 2001 -11;117(5):1091-1096.
Ståhle-Bäckdahl M, Inoue M, Guidice GJ, Parks WC. 92-kD gelatinase is produced by eosinophils at the site of blister formation in bullous pemphigoid and cleaves the extracellular domain of recombinant 180-kD bullous pemphigoid autoantigen. J Clin Invest 1994 -05;93(5):2022-2030.
Kelly EAB, Liu LY, Esnault S, Quinchia Johnson BH, Jarjour NN. Potent synergistic effect of IL-3 and TNF on matrix metalloproteinase 9 generation by human eosinophils. Cytokine 2012 -05;58(2):199-206.
Wakugawa M, Nakamura K, Hino H, Toyama K, Hattori N, Okochi H, et al. Elevated levels of eotaxin and interleukin-5 in blister fluid of bullous pemphigoid: correlation with tissue eosinophilia. Br J Dermatol 2000 -07;143(1):112-116.
Bonciani D, Quintarelli L, Del Bianco E, Bianchi B, Caproni M. Serum levels and tissue expression of interleukin-31 in dermatitis herpetiformis and bullous pemphigoid. J Dermatol Sci 2017 -08;87(2):210-212.
Rüdrich U, Gehring M, Papakonstantinou E, Illerhaus A, Engmann J, Kapp A, et al. Eosinophils are a Major Source of Interleukin-31 in Bullous Pemphigoid. Acta Derm Venereol 2018 -08-29;98(8):766-771.
Borradori L, Van Beek N, Feliciani C, Tedbirt B, Antiga E, Bergman R, et al. Updated S2 K guidelines for the management of bullous pemphigoid initiated by the European Academy of Dermatology and Venereology (EADV). J Eur Acad Dermatol Venereol 2022 -10;36(10):1689-1704.
Russo R, Capurro N, Cozzani E, Parodi A. Use of Dupilumab in Bullous Pemphigoid: Where Are We Now? J Clin Med 2022 -6-12;11(12):3367.
Abdat R, Waldman RA, de Bedout V, Czernik A, Mcleod M, King B, et al. Dupilumab as a novel therapy for bullous pemphigoid: A multicenter case series. J Am Acad Dermatol 2020 -07;83(1):46-52.
Zeng FAP, Murrell DF. Bullous pemphigoid—What do we know about the most recent therapies? Frontiers in medicine 2022 Nov 3,;9:1057096.
Simon D, Yousefi S, Cazzaniga S, Bürgler C, Radonjic S, Houriet C, et al. Mepolizumab failed to affect bullous pemphigoid: A randomized, placebo-controlled, double-blind phase 2 pilot study. Allergy 2020 -03;75(3):669-672.
Rhyou H, Han S, Nam Y. Successful induction treatment of bullous pemphigoid using reslizumab: a case report. Allergy Asthma Clin Immunol 2021 -11-16;17(1):117.
Dagher R, Kumar V, Copenhaver AM, Gallagher S, Ghaedi M, Boyd J, et al. Novel mechanisms of action contributing to benralizumab's potent anti-eosinophilic activity. Eur Respir J 2022 -03;59(3):2004306.
How to Cite
Downloads
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Artur Moraes-L'Abbate, Carolina Cortés Correa

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
| Article metrics | |
|---|---|
| Abstract views | |
| Galley vies | |
| PDF Views | |
| HTML views | |
| Other views | |