Dupilumab discontinuation in atopic dermatitis. Suspension may be an option? Follow-up of twelve cases
DOI:
https://doi.org/10.29176/2590843X.1679Palabras clave:
Antibodies, Atopic dermatitis, COVID-19, Dupilumab, Humanized, Monoclonal, TherapyResumen
Background:
Dupilumab is the first biologic treatment approved for moderate to severe atopic dermatitis (AD) with excellent security profile and efficacy but until the moment with little literature about its persistence.
Objective: To open the discussion and relevance of new studies about the possibility to suspend Dupilumab in some AD patients
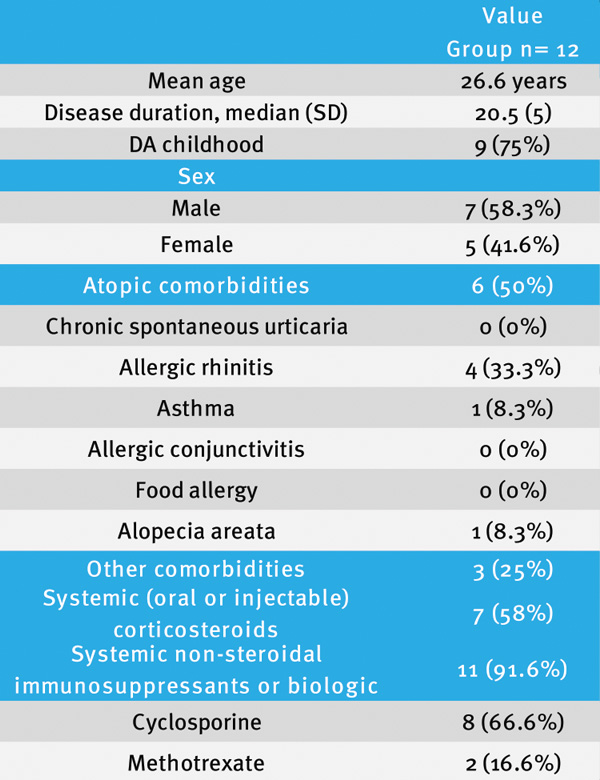
Methods: We conducted a retrospective review of the clinical charts from 101 AD patients that were receiving dupilumab. Eligible cases included patients who suspended treatment, regardless of the reason for discontinuation.
Results: Discontinuation was identified in 12 patients (11.8%), predominantly because of insurance (58.3%), adverse effects (8,3%), pregnancy (8,3%), no need of dupilumab(8,3%) and medication access difficulty during the COVID-19 pandemic( 8,3%).
After dupilumab suspension without restarting it 3 (33.3%) patients required immunomodulatory therapy and 1(11%) patient phototherapy;the remaining continued only with topical 8 (55%).
Conclusions: Our report opens the possibility for more studies about discontinuation of Dupilumab in AD patients who have some characteristics such as shorter course of the disease, less atopic comorbidities and less previous systemic immune-modulators.
Biografía del autor/a
Mirian Casas-Vargas, Cayre Clinical Center
Atopic Dermatitis Clinic, Department of Dermatology, Cayre Clinical Center, Bogotá, Colombia.
Cindy Cáceres-Salamanca, Foundation for Research in Dermatology
Foundation for Research in Dermatology - FUNINDERMA, Bogotá, Colombia.
María Fernanda Ordóñez-Rubiano, Central Military Hospital
Atopic Dermatitis Clinic, Department of Dermatology, Cayre Clinical Center. Division of Dermatology, Military Central Hospital, Bogotá, Colombia.
Referencias bibliográficas
Awosika O, Kim L, Mazhar M, Rengifo-Pardo M, Ehrlich A. Profile of dupilumab and its potential in the treatment of inadequately controlled moderate-to-severe atopic dermatitis. Clin Cosmet Investig Dermatol. 2018 Jan;Volume 11:41–9.DOI: https://doi.org/10.2147/CCID.S123329
Renert-Yuval Y, Guttman-Yassky E. New treatments for atopic dermatitis targeting beyond IL-4/IL-13 cytokines. Ann Allergy Asthma Immunol Off Publ Am Coll Allergy Asthma Immunol. 2020 Jan;124(1):28–35. DOI: https://doi.org/10.1016/j.anai.2019.10.005
Damiani G, Calzavara‐Pinton P, Stingeni L, Hansel K, Cusano F, “Skin Allergy” Group of SIDeMaST, et al. Italian guidelines for therapy of atopic dermatitis—Adapted from consensus‐based European guidelines for treatment of atopic eczema (atopic dermatitis). Dermatol Ther [Internet]. 2019 Nov [cited 2021 Feb 21];32(6). DOI: https://doi.org/10.1111/dth.13121
Ariëns LFM, Bakker DS, van der Schaft J, Garritsen FM, Thijs JL, de Bruin-Weller MS. Dupilumab in atopic dermatitis: rationale, latest evidence and place in therapy. Ther Adv Chronic Dis. 2018 Sep;9(9):159–70.DOI: https://doi.org/10.1177/2040622318773686
Beck LA, Thaçi D, Deleuran M, Blauvelt A, Bissonnette R, de Bruin-Weller M, et al. Dupilumab Provides Favorable Safety and Sustained Efficacy for up to 3 Years in an Open-Label Study of Adults with Moderate-to-Severe Atopic Dermatitis. Am J Clin Dermatol. 2020;21(4):567–77.DOI: https://doi.org/10.1007/s40257-020-00527-x
Iuga AO, McGuire MJ. Adherence and health care costs. Risk Manag Healthc Policy. 2014 Feb 20;7:35–44.DOI: https://doi.org/10.2147/RMHP.S19801
Osterberg L, Blaschke T. Adherence to Medication. N Engl J Med. 2005 Aug 4;353(5):487–97.DOI: https://doi.org/10.1056/NEJMra050100
Blauvelt A, Bruin-Weller M de, Gooderham M, Cather JC, Weisman J, Pariser D, et al. Long-term management of moderate-to-severe atopic dermatitis with dupilumab and concomitant topical corticosteroids (LIBERTY AD CHRONOS): a 1-year, randomised, double-blinded, placebo-controlled, phase 3 trial. The Lancet. 2017 Jun 10;389(10086):2287–303.DOI: https://doi.org/10.1016/S0140-6736(17)31191-1
Treister AD, Kraff-Cooper C, Lio PA. Risk Factors for Dupilumab-Associated Conjunctivitis in Patients With Atopic Dermatitis. JAMA Dermatol. 2018 Oct;154(10):1208–11.DOI: https://doi.org/10.1001/jamadermatol.2018.2690
Ordóñez‐Rubiano MF, Campo I, Casas M. Dupilumab in atopic dermatitis, a protocol for SARS-COV-2-infected patients. Dermatol Ther. 2020;33(6):e14172. DOI: https://doi.org/10.1111/dth.14172
Hamilton JD, Suárez-Fariñas M, Dhingra N, Cardinale I, Li X, Kostic A, et al. Dupilumab improves the molecular signature in skin of patients with moderate-to-severe atopic dermatitis. J Allergy Clin Immunol. 2014 Dec;134(6):1293–300.DOI: https://doi.org/10.1016/j.jaci.2014.10.013
Silverberg JI, Guttman-Yassky E, Gadkari A, Kuznik A, Mallya UG, Mastey V, et al. Real-world persistence with dupilumab among adults with atopic dermatitis. Ann Allergy Asthma Immunol. 2021 Jan;126(1):40–5.DOI: https://doi.org/10.1016/j.anai.2020.07.026
Cómo citar
Descargas
Publicado
Cómo citar
Número
Sección
Licencia
Derechos de autor 2024 Mirian Casas-Vargas, Cindy Cáceres-Salamanca, María Fernanda Ordóñez-Rubiano

Esta obra está bajo una licencia internacional Creative Commons Atribución-NoComercial-CompartirIgual 4.0.
| Estadísticas de artículo | |
|---|---|
| Vistas de resúmenes | |
| Vistas de PDF | |
| Descargas de PDF | |
| Vistas de HTML | |
| Otras vistas | |