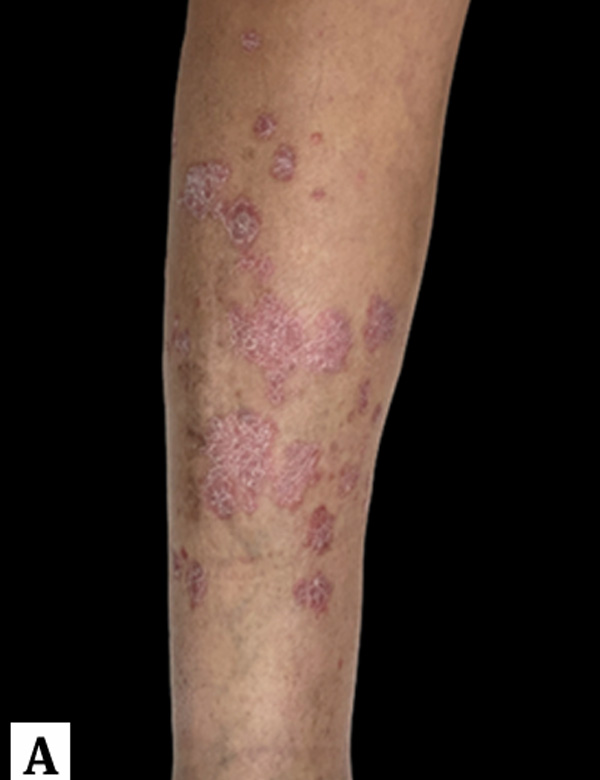
Eventos adversos cutáneos relacionados con inhibidores de puntos de control inmunológico: una revisión
DOI:
https://doi.org/10.29176/2590843X.1928Palabras clave:
inmunoterapia, Inhibidores de Puntos de Control Inmunológico, Inhibidor CTLA-4, Inhibidor PD-1, Inhibidor PD-L1, Eventos Adversos Inmunorelacionados, Erupciones por MedicamentosResumen
Los inhibidores de puntos de control inmunológico (ICPi por su sigla en inglés) son fármacos novedosos aprobados para el tratamiento de diferentes tumores malignos en estadios avanzados y su uso se ha incrementado dado que se trata de una de las estrategias más efectivas al demostrar una mejoría en la supervivencia general. No obstante, los eventos adversos relacionados con la activación del sistema inmunológico son frecuentes, dentro de los que se destaca un amplio abanico de reacciones mucocutáneas de diferentes grados de severidad. Un enfoque inicial basado en la identificación temprana de la toxicidad cutánea y la evaluación de la gravedad permitirá determinar el nivel de complejidad asistencial que el paciente requiere, las medidas terapéuticas específicas y la evaluación de la interrupción o no del ICPi. En la siguiente revisión narrativa se explica el funcionamiento de los ICPi, las toxicidades cutáneas y el enfoque para el manejo de las mismas.
Biografía del autor/a
Angela María Lara Botina, Hospital de Infecciosas Francisco Javier Muñiz, Ciudad Autónoma de Buenos Aires, Argentina
Médica especialista en dermatología. Servicio de Dermatología, Hospital de Infecciosas Francisco Javier Muñiz, Ciudad Autónoma de Buenos Aires, Argentina.
Olga Lucía Forero, Hospital de Infecciosas Francisco Javier Muñiz, Ciudad Autónoma de Buenos Aires, Argentina
Médica especialista en dermatología. Médica de planta. Servicio de Dermatología, Hospital de Infecciosas Francisco Javier Muñiz, Ciudad Autónoma de Buenos Aires, Argentina.
María Emilia Candiz, Hospital de Infecciosas Francisco Javier Muñiz, Ciudad Autónoma de Buenos Aires, Argentina
Médica especialista en Dermatología. Médica de planta. Servicio de Dermatología, Hospital de Infecciosas Francisco Javier Muñiz, Ciudad Autónoma de Buenos Aires, Argentina.
Viviana Leiro, Hospital de Infecciosas Francisco Javier Muñiz, Ciudad Autónoma de Buenos Aires, Argentina
Jefa de unidad, servicio de dermatología. Servicio de Dermatología, Hospital de Infecciosas Francisco Javier Muñiz, Ciudad Autónoma de Buenos Aires, Argentina.
Esteban Maronna, Hospital de Infecciosas Francisco Javier Muñiz, Ciudad Autónoma de Buenos Aires, Argentina
Médico especialista en dermatopatología. Servicio de Dermatología, Hospital de Infecciosas Francisco Javier Muñiz, Ciudad Autónoma de Buenos Aires, Argentina.
Referencias bibliográficas
Watanabe T, Yamaguchi Y. Cutaneous manifestations associated with immune checkpoint inhibitors. Front Immunol. 2023;14:1071983. http://doi.org/10.3389/fimmu.2023.1071983. PMID: 36891313; PMCID: PMC9986601
Juan-Carpena G, Palazón-Cabanes JC, Blanes-Martínez M. Characteristics, Management, and Prognostic Implications of Adverse Effects of Immune Checkpoint Inhibitors: A Systematic Review. Actas Dermosifiliogr. 2022;113(4):376-87. http://doi.org/10.1016/j.ad.2021.09.005. Epub 2021 Dec 31. PMID: 35623728.
Postow MA, Sidlow R, Hellmann MD. Immune-Related Adverse Events Associated with Immune Checkpoint Blockade. N Engl J Med. 2018;378(2):158-68. http://doi.org/10.1056/NEJMra1703481. PMID: 29320654.
Coleman E, Ko C, Dai F, Tomayko MM, Kluger H, Leventhal JS. Inflammatory eruptions associated with immune checkpoint inhibitor therapy: A single-institution retrospective analysis with stratification of reactions by toxicity and implications for management. J Am Acad Dermatol. 2019;80(4):990-997. http://doi.org/10.1016/j.jaad.2018.10.062. Epub 2018 Nov 3. PMID: 30399387; PMCID: PMC6420863
Gault A, Anderson AE, Plummer R, Stewart C, Pratt AG, Rajan N. Cutaneous immune-related adverse events in patients with melanoma treated with checkpoint inhibitors. Br J Dermatol. 2021;185(2):263-271. http://doi.org/10.1111/bjd.19750. Epub 2021 Feb 1. PMID: 33393076
Patel AB, Farooq S, Welborn M, Amaria RN, Chon SY, Diab A et al. Cutaneous adverse events in 155 patients with metastatic melanoma consecutively treated with anti-CTLA4 and anti-PD1 combination immunotherapy: Incidence, management, and clinical benefit. Cancer. 2022;128(5):975-983. http://doi.org/10.1002/cncr.34004. Epub 2021 Nov 1. PMID: 34724197.
Barquín-García A, Molina-Cerrillo J, Garrido P, Garcia-Palos D, Carrato A, Alonso-Gordoa T. New oncologic emergencies: What is there to know about inmunotherapy and its potential side effects? Eur J Intern Med. 2019;66:1-8. http://doi.org/10.1016/j.ejim.2019.05.020. Epub 2019 May 23. PMID: 31130304.
Reyes SJ, González KB, Rodríguez C, Rodríguez C, Navarrete-Muñoz C, Salazar AP et al. Actualización general de inmunoterapia en cáncer. Rev. méd. 2020;148(7):970-82. http://dx.doi.org/10.4067/S0034-98872020000700970 Disponible en: http://www.scielo.cl/scielo.php?script=sci_arttext&pid=S0034-98872020000700970&lng=es.
Tamariz-Campillo AF, Corona-Deschamps A, Mellado-Orellana R, Diaz-Greene EJ, Rodríguez-Weber FL. Inhibidores de puntos de control inmunitario en estadios avanzados de melanoma. Acta Med. 2021; 19(1):108-15. https://dx.doi.org/10.35366/98579
Topalian SL, Taube JM, Anders RA, Pardoll DM. Mechanism-driven biomarkers to guide immune checkpoint blockade in cancer therapy. Nat Rev Cancer. 2016;16(5):275-87. http://doi.org/10.1038/nrc.2016.36. Epub 2016 Apr 15. PMID: 27079802; PMCID: PMC5381938
Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252-64. http://doi.org/10.1038/nrc3239
Sibaud V. Dermatologic Reactions to Immune Checkpoint Inhibitors: Skin Toxicities and Immunotherapy. Am J Clin Dermatol. 2018;19(3):345-361. http://doi.org/10.1007/s40257-017-0336-3. PMID: 29256113
Molinero-Muñoz M, Zamora-Barrios MD, Martínez-Sesmero JM. Efectividad y seguridad de inhibidores de puntos de control inmunitario en pacientes excluidos de ensayos clínicos. Rev. OFIL·ILAPHAR [Internet]. 2020 Dic [citado 2023 Dic 04]; 30(4):325-28. Disponible en: http://scielo.isciii.es/scielo.php?script=sci_arttext&pid=S1699-714X2020000400010&lng=es.
Quach HT, Johnson DB, LeBoeuf NR, Zwerner JP, Dewan AK. Cutaneous adverse events caused by immune checkpoint inhibitors. J Am Acad Dermatol. 2021;85(4):956-966. http://doi.org/10.1016/j.jaad.2020.09.054. Epub 2021 Jul 28. PMID: 34332798
Goldinger SM, Stieger P, Meier B, Micaletto S, Contassot E, French LE et al Cytotoxic Cutaneous Adverse Drug Reactions during Anti-PD-1 Therapy. Clin Cancer Res. 2016;22(16):4023-9. http://doi.org/10.1158/1078-0432.CCR-15-2872. Epub 2016 Mar 8. PMID: 26957557
Muhaj F, Karri PV, Moody W, Brown A, Patel AB. Mucocutaneous adverse events to immune checkpoint inhibitors. Front Allergy. 2023;4:1147513. http://doi.org/10.3389/falgy.2023.1147513. PMID: 36938327; PMCID: PMC10017442.
Lema-Medina M, Llinás N, Bravo A, Hernández JC. Nuevos registros sanitarios en oncología en Colombia durante la pandemia COVID-19. Parte 2: inhibidores de puntos de control inmunológico. Rev.Col.Hematol.Oncol [Internet]. 3 de agosto de 2023 [citado 4 de diciembre de 2023];10(1). Disponible en: https://revista.acho.info/index.php/acho/article/view/408
Postow M, Johnson DB. Toxicities associated with immune checkpoint inhibitors. In: UpToDate, Atkins MB (Ed), UpToDate, Waltham, MA. (citado citado 4 de diciembre de 2023.)
Cutroneo P, Ingrasciotta Y, Isgrò V, Rullo EV, Berretta M, Fiorica F et al. Psoriasis and psoriasiform reactions secondary to immune checkpoint inhibitors. Dermatol Ther. 2021;34(2):e14830. http://doi.org/10.1111/dth.14830. Epub 2021 Feb 11. PMID: 33527643
Apalla Z, Nikolaou V, Fattore D, Fabbrocini G, Freites-Martinez A, Sollena P et al. European recommendations for management of immune checkpoint inhibitors-derived dermatologic adverse events. The EADV task force 'Dermatology for cancer patients' position statement. J Eur Acad Dermatol Venereol. 2022;36(3):332-350. http://doi.org/10.1111/jdv.17855. Epub 2021 Dec 15. PMID: 34910332
Tsiogka A, Bauer JW, Patsatsi A. Bullous Pemphigoid Associated with Anti-programmed Cell Death Protein 1 and Anti-programmed Cell Death Ligand 1 Therapy: A Review of the Literature. Acta Derm Venereol. 2021;20;101(1):adv00377. http://doi.org/10.2340/00015555-3740. PMID: 33426566; PMCID: PMC9309843
Hasan Ali O, Berner F, Bomze D, Fässler M, Diem S, Cozzio A et al. Human leukocyte antigen variation is associated with adverse events of checkpoint inhibitors. Eur J Cancer. 2019;107:8-14. http://doi.org/10.1016/j.ejca.2018.11.009. Epub 2018 Dec 7. PMID: 30529903
Apalla Z, Papageorgiou C, Lallas A, Delli F, Fotiadou C, Kemanetzi C et al. Cutaneous Adverse Events of Immune Checkpoint Inhibitors: A Literature Review. Dermatol Pract Concept. 2021;11(1):e2021155. http://doi.org/10.5826/dpc.1101a155. PMID: 33614223; PMCID: PMC7875661
Schneider BJ, Naidoo J, Santomasso BD, Lacchetti C, Adkins S, Anadkat M et al. Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: ASCO Guideline Update. J Clin Oncol. 2021;39(36):4073-4126. http://doi.org/10.1200/JCO.21.01440. Epub 2021 Nov 1. Erratum in: J Clin Oncol. 2022 Jan 20;40(3):315. PMID: 34724392
Patel AB, Pacha O. Skin Reactions to Immune Checkpoint Inhibitors. Adv Exp Med Biol. 2020;1244:235-246. http://doi.org/10.1007/978-3-030-41008-7_11. PMID: 32301018
Geisler AN, Phillips GS, Barrios DM, Wu J, Leung DYM, Moy AP et al. Immune checkpoint inhibitor-related dermatologic adverse events. J Am Acad Dermatol. 2020;83(5):1255-1268. http://doi.org/10.1016/j.jaad.2020.03.132. Epub 2020 May 23. PMID: 32454097; PMCID: PMC7572894.
Patel AB. Cutaneous immune-related adverse events associated with immune checkpoint inhibitors. In: UpToDate, Mockenhaupt M, Atkins MB, (Ed), UpToDate. Waltham, MA. (citado en enero 25 de 2024.)
Malviya N, Tattersall IW, Leventhal J, Alloo A. Cutaneous immune-related adverse events to checkpoint inhibitors. Clin Dermatol. 2020;38(6):660-678. http://doi.org/10.1016/j.clindermatol.2020.06.011. Epub 2020 Jun 27. PMID: 33341200.
Nikolaou V, Sibaud V, Fattore D, Sollena P, Ortiz-Brugués A, Giacchero D et al. Immune checkpoint-mediated psoriasis: A multicenter European study of 115 patients from the European Network for Cutaneous Adverse Event to Oncologic Drugs (ENCADO) group. J Am Acad Dermatol. 2021;84(5):1310-1320. http://doi.org/10.1016/j.jaad.2020.08.137. Epub 2020 Dec 3. PMID: 33279646.
Cutroneo P, Ingrasciotta Y, Isgrò V, Rullo EV, Berretta M, Fiorica F et al. Psoriasis and psoriasiform reactions secondary to immune checkpoint inhibitors. Dermatol Ther. 2021;34(2):e14830. http://doi.org/10.1111/dth.14830. Epub 2021 Feb 11. PMID: 33527643
Siciliano MA, Dastoli S, d'Apolito M, Staropoli N, Tassone P, Tagliaferri P et al. Pembrolizumab-Induced Psoriasis in Metastatic Melanoma: Activity and Safety of Apremilast, a Case Report. Front Oncol. 2020;10:579445. http://doi.org/10.3389/fonc.2020.579445. PMID: 33163407; PMCID: PMC7591674.
Halle BR, Betof Warner A, Zaman FY, Haydon A, Bhave P, Dewan AK et al. Immune checkpoint inhibitors in patients with pre-existing psoriasis: safety and efficacy. J Immunother Cancer. 2021;9(10):e003066. http://doi.org/10.1136/jitc-2021-003066. PMID: 34635495; PMCID: PMC8506877
Sahuquillo-Torralba A, Ballester-Sánchez R, Pujol-Marco C, Botella-Estrada R. Pembrolizumab: a new Drug That Can Induce Exacerbations of Psoriasis. Actas Dermosifiliogr. 2016;107(3):264-6. English, Spanish. http://doi.org/10.1016/j.ad.2015.07.012. Epub 2015 Nov 3. PMID: 26546027
Kratzsch D, Simon JC, Ziemer M. Lichen planus-like drug eruption on anti-PD-1 therapy. J Dtsch Dermatol Ges. 2017;15(12):1238-1240. http://doi.org/10.1111/ddg.13370. Epub 2017 Dec 6. PMID: 29211335
Shi VJ, Rodic N, Gettinger S, Leventhal JS, Neckman JP, Girardi M et al. Clinical and Histologic Features of Lichenoid Mucocutaneous Eruptions Due to Anti-Programmed Cell Death 1 and Anti-Programmed Cell Death Ligand 1 Immunotherapy. JAMA Dermatol. 2016;152(10):1128-1136. http://doi.org/10.1001/jamadermatol.2016.2226. PMID: 27411054; PMCID: PMC6108080
Ravi V, Maloney NJ, Worswick S. Neutrophilic dermatoses as adverse effects of checkpoint inhibitors: A review. Dermatol Ther. 2019;32(5):e13074. http://doi.org/10.1111/dth.13074. Epub 2019 Sep 4. PMID: 31444856
Welborn ME, Kubicki SL, Patel AB. Pyoderma gangrenosum following initiation of immune checkpoint inhibitor therapy. Journal of Immunotherapy and Precision Oncology. 2018;1(2):82-84. http://doi.org/10.4103/JIPO.JIPO_11_18
Siegel J, Totonchy M, Damsky W, Berk-Krauss J, Castiglione F Jr, Sznol M et al. Bullous disorders associated with anti-PD-1 and anti-PD-L1 therapy: A retrospective analysis evaluating the clinical and histopathologic features, frequency, and impact on cancer therapy. J Am Acad Dermatol. 2018;79(6):1081-1088. http://doi.org/10.1016/j.jaad.2018.07.008. Epub 2018 Jul 17. PMID: 30025829.
Sadik CD, Langan EA, Gutzmer R, Fleischer MI, Loquai C, Reinhardt L et al. Retrospective Analysis of Checkpoint Inhibitor Therapy-Associated Cases of Bullous Pemphigoid From Six German Dermatology Centers. Front Immunol. 2021;11:588582. http://doi.org/10.3389/fimmu.2020.588582. PMID: 33708189; PMCID: PMC7940359
Kwon CW, Murthy RK, Kudchadkar R, Stoff BK. Pembrolizumab-induced lichen planus pemphigoides in a patient with metastatic Merkel cell carcinoma. JAAD Case Rep. 2020;6(10):1045-1047. http://doi.org/10.1016/j.jdcr.2020.03.007. PMID: 32995442; PMCID: PMC7509362.
Schwartzman G, Simpson MM, Jones R, Schiavone K, Coffman M, Meyerle J. Anti-PD1 immune checkpoint inhibitor-induced bullous pemphigoid in metastatic melanoma and non-small cell lung cancer. Cutis. 2020;105(6):E9-E12. PMID: 32716997.
Chen WS, Tetzlaff MT, Diwan H, Jahan-Tigh R, Diab A, Nelson K et al. Suprabasal acantholytic dermatologic toxicities associated checkpoint inhibitor therapy: A spectrum of immune reactions from paraneoplastic pemphigus-like to Grover-like lesions. J Cutan Pathol. 2018;45(10):764-773. http://doi.org/10.1111/cup.13312. Epub 2018 Jul 24. PMID: 29943453.
Maloney NJ, Ravi V, Cheng K, Bach DQ, Worswick S. Stevens-Johnson syndrome and toxic epidermal necrolysis-like reactions to checkpoint inhibitors: a systematic review. Int J Dermatol. 2020;59(6):e183-e188. http://doi.org/10.1111/ijd.14811. Epub 2020 Feb 13. PMID: 32052409
Coleman EL, Olamiju B, Leventhal JS. The life-threatening eruptions of immune checkpoint inhibitor therapy. Clin Dermatol. 2020;38(1):94-104. http://doi.org/10.1016/j.clindermatol.2019.10.015. Epub 2019 Oct 25. PMID: 32197753.
Lu J, Thuraisingam T, Chergui M, Nguyen K. Nivolumab-associated DRESS syndrome: A case report. JAAD Case Rep. 2019;5(3):216-218. http://doi.org/10.1016/j.jdcr.2018.11.017. PMID: 30809563; PMCID: PMC6374958.
Mirza S, Hill E, Ludlow SP, Nanjappa S. Checkpoint inhibitor-associated drug reaction with eosinophilia and systemic symptom syndrome. Melanoma Res. 2017;27(3):271-273. http://doi.org/10.1097/CMR.0000000000000326. PMID: 28146044
Hwang SJ, Carlos G, Wakade D, Sharma R, Fernandez-Penas P. Ipilimumab-induced acute generalized exanthematous pustulosis in a patient with metastatic melanoma. Melanoma Res. 2016;26(4):417-20. http://doi.org/10.1097/CMR.0000000000000261. PMID: 27031538.
Guida M, Strippoli S, Maule M, Quaglino P, Ramondetta A, Chiaron Sileni V et al. Immune checkpoint inhibitor associated vitiligo and its impact on survival in patients with metastatic melanoma: an Italian Melanoma Intergroup study. ESMO Open. 2021;6(2):100064. http://doi.org/10.1016/j.esmoop.2021.100064. Epub 2021 Mar 10. PMID: 33711672; PMCID: PMC7970061.
Allenbach Y, Anquetil C, Manouchehri A, Benveniste O, Lambotte O, Lebrun-Vignes B et al. Immune checkpoint inhibitor-induced myositis, the earliest and most lethal complication among rheumatic and musculoskeletal toxicities. Autoimmun Rev. 2020;19(8):102586. http://doi.org/10.1016/j.autrev.2020.102586. Epub 2020 Jun 11. PMID: 32535094
Michot JM, Fusellier M, Champiat S, Velter C, Baldini C, Voisin AL et al. Drug-induced lupus erythematosus following immunotherapy with anti-programmed death-(ligand) 1. Ann Rheum Dis. 2019;78(7):e67. http://doi.org/10.1136/annrheumdis-2018-213677. Epub 2018 Jun 1. PMID: 29858173.
Blakeway EA, Elshimy N, Muinonen-Martin A, Marples M, Mathew B, Mitra A. Cutaneous lupus associated with pembrolizumab therapy for advanced melanoma: a report of three cases. Melanoma Res. 2019;29(3):338-341. http://doi.org/10.1097/CMR.0000000000000587. PMID: 30762712
Guerra NL, Matas-García A, Serra-García L, Morgado-Carrasco D, Padrosa J, Aldecoa I et al. Dermatomyositis unleashed by immune checkpoint inhibitors. Three additional cases and a review of the literature. Autoimmun Rev. 2023;22(8):103375. http://doi.org/10.1016/j.autrev.2023.103375. Epub 2023 Jun 13. PMID: 37321468; PMCID: PMC10529928
Terrier B, Humbert S, Preta LH, Delage L, Razanamahery J, Laurent-Roussel S et al. Risk of scleroderma according to the type of immune checkpoint inhibitors. Autoimmun Rev. 2020;19(8):102596. http://doi.org/10.1016/j.autrev.2020.102596. Epub 2020 Jun 12. PMID: 32540450
Higashi T, Miyamoto H, Yoshida R, Furuta Y, Nagaoka K, Naoe H et al. Sjögren's Syndrome as an Immune-related Adverse Event of Nivolumab Treatment for Gastric Cancer. Intern Med. 2020;59(20):2499-2504. http://doi.org/10.2169/internalmedicine.4701-20. Epub 2020 Jun 23. PMID: 32581160; PMCID: PMC7662059
Lacouture M, Sibaud V. Toxic Side Effects of Targeted Therapies and Immunotherapies Affecting the Skin, Oral Mucosa, Hair, and Nails. Am J Clin Dermatol. 2018;19(Suppl 1):31-39. http://doi.org/10.1007/s40257-018-0384-3. PMID: 30374901; PMCID: PMC6244569
Schneider BJ, Naidoo J, Santomasso BD, Lacchetti C, Adkins S, Anadkat M et al. Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: ASCO Guideline Update. J Clin Oncol. 2021;39(36):4073-4126. http://doi.org/10.1200/JCO.21.01440. Epub 2021 Nov 1. Erratum in: J Clin Oncol. 2022 Jan 20;40(3):315. PMID: 34724392.
Thompson JA, Schneider BJ, Brahmer J, Achufusi A, Armand P, Berkenstock MK et al. Management of Immunotherapy-Related Toxicities, Version 1.2022, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2022;20(4):387-405. http://doi.org/10.6004/jnccn.2022.0020. PMID: 35390769.
Haanen JBAG, Carbonnel F, Robert C, Kerr KM, Peters S, Larkin J et al; ESMO Guidelines Committee. Management of toxicities from immunotherapy: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017;28(suppl_4):iv119-iv142. http://doi.org/10.1093/annonc/mdx225. Erratum in: Ann Oncol. 2018 Oct 1;29(Suppl 4):iv264-iv266. PMID: 28881921.
Puzanov I, Diab A, Abdallah K, Bingham CO, Brogdon C, Dadu R et al. Society for Immunotherapy of Cancer Toxicity Management Working Group. Managing toxicities associated with immune checkpoint inhibitors: consensus recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. J Immunother Cancer. 2017;5(1):95. http://doi.org/10.1186/s40425-017-0300-z. PMID: 29162153; PMCID: PMC5697162.
Choi J, Anderson R, Blidner A, Cooksley T, Dougan M, Glezerman I et al. Multinational Association of Supportive Care in Cancer (MASCC) 2020 clinical practice recommendations for the management of severe dermatological toxicities from checkpoint inhibitors. Support Care Cancer. 2020;28(12):6119-6128. http://doi.org/10.1007/s00520-020-05706-4. Epub 2020 Aug 27. PMID: 32856211; PMCID: PMC8996262.
Cómo citar
Descargas
Publicado
Cómo citar
Número
Sección
Licencia
Derechos de autor 2025 Angela María Lara Botina, Olga Lucía Forero, María Emilia Candiz, Viviana Leiro, Esteban Maronna

Esta obra está bajo una licencia internacional Creative Commons Atribución-NoComercial-CompartirIgual 4.0.
| Estadísticas de artículo | |
|---|---|
| Vistas de resúmenes | |
| Vistas de PDF | |
| Descargas de PDF | |
| Vistas de HTML | |
| Otras vistas | |